La Jolla, California based toSense has received of 510(k) clearance from the Food and Drug Administration (FDA) for the company’s CoVa Monitoring System (CoVa), a novel body-worn sensor for at-home patients with chronic illnesses, such as congestive heart failure (CHF), chronic obstructive pulmonary disease (COPD), hypertension, and renal failure.
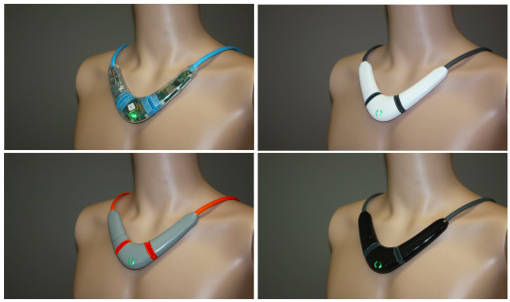
CoVa provides clinical-grade diagnostics remotely monitoring thoracic impedance, heart rate, heart rate variability, respiration rate, skin temperature, and posture. Worn like a conventional necklace for just minutes each day, the CoVa Sensor measures bioimpedance and electrocardiogram waveforms, and from these data is able to calculate the patient’s health status in near real time. Electronics that measure the monitored parameters are embedded within the Sensor, resulting in a comfortable, easy-to-use device with no external wires.
The Sensor, worn like a conventional necklace, drapes around the patient’s neck and turns on with a simple magnetic clasp with magnetic electrodes that attaches to the patient’s skin. Once the Sensor is on, it takes less than five minutes to compete its measurements.
The Sensor is designed for older patients, emphasizing usability with such features as the magnetic clasp to turn the Sensor on, magnetic electrodes, and simple wireless data transmission. CoVa is designed for use in the home environment by elderly patients, enabling medical professionals to monitor and review data remotely. Several gateways (see below) can be employed to forward data to the Cloud, and collectively these tools make it possible for patients to be monitored in the comfort of their own homes.
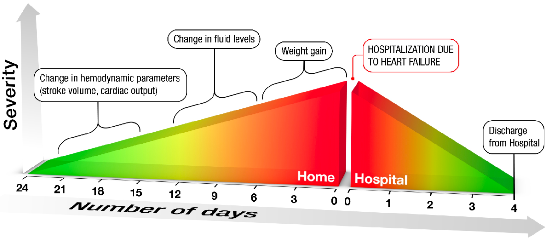
A goal of the CoVa Monitoring System is to make at-home monitoring of cardiopulmonary parameters for heart failure patients as simple as glucose monitoring for diabetics. Current solutions are relegated to technologies such as weight scales, which are insensitive to physiological parameters and are unable to characterize early onset of CHF. Weight is a late indicator of a potential heart failure event. As shown in the chart below, measurements such as fluid level, stroke volume, and cardiac output provide an early indication of a potential heart failure event. With the CoVa Monitoring System, this information is available remotely to physicians and caregivers.
“Heart failure is the most important cause of hospital admission and readmission in the United States.” says Dr. Eric Topol, Chief Academic
Officer of Scripps Health, a consultant cardiologist practitioner at Scripps Clinic, a professor of genomics at The Scripps Research Institute, and director of the Scripps Translational Science Institute — a National Institutes of Health funded program of the Clinical and Translational Science Award (CTSA) Consortium focused on advancing individualized medicine, and an advisor for toSense. “This innovative necklace technology to track chest fluid status and key heart parameters has promise for use in patients with heart failure.” Recently, Dr. Topol was selected as one of the “100 Most Influential People in Healthcare” in addition to being named the No. 1 “Physician Executive” in the nation, both by Modern Healthcare.
Research at the Translational Science Institute Topol lab is focused on individualized medicine, using the genome and digital technologies to understand each person at the biologic, physiologic granular level to determine appropriate therapies and prevention. An example is the use of pharmacogenomics and the lab’s research on clopidogrel (Plavix). By determining the reasons for why such a large proportion of people do not respond to this medication, researchers can use alternative treatment strategies to prevent blood clots.
“CoVa will be transformational in delivering home-health monitoring. Clinicians will have remote access to important, actionable data.” says toSense CTO Matt Banet. “The system’s low price point allows universal access to a new, exciting sensor that could be a critical component of the healthcare system.”
CoVa is indicated for patients: i) with fluid-management problems; ii) taking diuretic medication; iii) living with heart failure; iv) living with end-stage renal disease; v) recovering from a coronary artery disease-related event; and/or vi) suffering from recurrent dehydration. Currently the CoVa Monitoring System requires a prescription. Future versions will be available to consumers.
The CoVa Monitoring System has three components: the Sensor, a Gateway, and a Web-based system. The Sensor measures simple vital signs as well as complex hemodynamic parameters. The system supports several Gateways and Web-based systems. Once the Sensor has completed its measurements, information is sent wirelessly via Bluetooth to a Gateway.
The Gateway collects the data gathered by the Sensor and forwards it to a Web-based system. Currently, there are two Gateway options: an Android device, such as a tablet or smart phone, and Qualcomm’s 2net System. The Android device allows users to view current measurements and historical data. The 2net System simply plugs into a wall outlet and requires no user interaction. For both Gateways, data is automatically forwarded to the Web-based system when the measurement is complete.
Several Web-based systems, supporting a variety of monitoring experiences, are also integrated with the CoVa Monitoring System. A clinical professional can log on to the Web-based system to monitor a patient’s health. With these tools, clinical providers can easily identify patients that may need to modify medications, exercise, or diet to avoid a potential hospitalizations.
For more information about toSense visit:
http://www.toSense.com
Sources:
toSense
Scripps Health
Image Credits:
toSense
Scripps Health