 Researchers at the University of Louisville have launched a research trial to study a proprietary investigational medical device called the “Zephyr Endobronchial Valve” (EBV) that is designed to help patients with emphysema by shutting off diseased areas of the lung. UofL is the only site in Kentucky among 14 sites nationwide testing the device.
Researchers at the University of Louisville have launched a research trial to study a proprietary investigational medical device called the “Zephyr Endobronchial Valve” (EBV) that is designed to help patients with emphysema by shutting off diseased areas of the lung. UofL is the only site in Kentucky among 14 sites nationwide testing the device.
Emphysema is a chronic, progressive disorder of the lower respiratory tract and a type of chronic obstructive pulmonary disease (COPD). It is seriously disabling with potential for major complications and is often terminal. Emphysema symptoms include shortness of breath and wheezing in the lungs during respiration. The primary causes of the disease are smoking and/or other long-term exposures to inhaled irritants including air pollution, chemicals, manufacturing fumes, or airborne small particles such as coal dust.
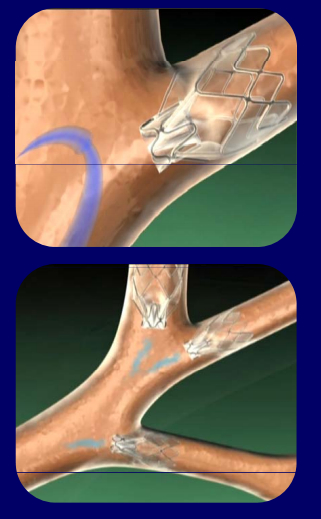
 The Zephyr Endobronchial Valve (EBV) is a one-way valve that blocks off diseased sections of the lung to inhaled air, while still allowing trapped air already inside the lung to escape. This enables the the diseased part of the lung to collapse and allows for healthier parts of the lung to expand and function more efficiently.
The Zephyr Endobronchial Valve (EBV) is a one-way valve that blocks off diseased sections of the lung to inhaled air, while still allowing trapped air already inside the lung to escape. This enables the the diseased part of the lung to collapse and allows for healthier parts of the lung to expand and function more efficiently.
The manufacturer of the EBV device, Pulmonx Inc. of Redwood City, California, cites advantages of the EBV that include broad clinical experience of any minimally invasive emphysema treatment over a decade of clinical experience with more than 25,000 valves having ben implanted.
More than 60 publications have documented the EBV’s safety, efficacy and long-term survival benefits for patients treated with severe emphysema. Patients experience a significant improvement in lung function, exercise tolerance, and quality of life, as well as long-term survival benefits. Pulmonx cites significantly improved FEV and sustained functional outcomes, clinically significant improvement in exercise tolerance, and improved long-term survival through five years.

The randomized study, which is sponsored by Pulmonx Inc. and known as the LIBERATE study, is to investigate the EBV’s safety and effectiveness for use in treating emphysema symptoms compared with a current standard medical therapy program alone. Principal investigator of the UofL site study is Tanya Wiese, D.O. , Assistant Professor of Medicine sand director of the Interventional Pulmonary Program at the University of Louisville Health Care Outpatient Center.
“The Zephyr EBV’s novel mechanism of action shows promise to help the healthy parts of the lung expand and reduce the effect of the disease,” observes Dr. Weise. “While not a cure, we believe this device could bring relief and improved quality of life to our patients with emphysema.”
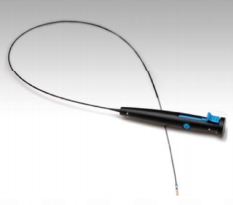 A surgeon can implant the EBV in a diseased section of a patient’s lungs using bronchoscopy, a procedure in which the lungs are accessed via a small tube with a camera mounted on its tip. Employing bronchoscopy, a physician can reach the airways in the lung by passing the tube through either the mouth or nose so invasive thoracic surgery is not required.
A surgeon can implant the EBV in a diseased section of a patient’s lungs using bronchoscopy, a procedure in which the lungs are accessed via a small tube with a camera mounted on its tip. Employing bronchoscopy, a physician can reach the airways in the lung by passing the tube through either the mouth or nose so invasive thoracic surgery is not required.
[adrotate group=”3″]
The UoL release notes incidence of emphysema is particularly acute in Kentucky. According to American Lung Association estimates, more than 56,000 Kentuckians, or a startling 13 percent of the state’s population, have emphysema, one of the highest rates of any region of the United States.
Other trial LIBERATE trial sites are Arizona Pulmonary Specialists, Cleveland Clinic, Duke University Medical Center, Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Southern Illinois University School of Medicine, Temple University Hospital, University of Alabama at Birmingham, Beth Israel Deaconess Medical Center, University of Pittsburg Medical Center, The Mayo Clinic, University of California at Davis Medical Center, University of California, San Francisco and University of Southern Enrollment.
Enrolment in the UoL study is expected to be complete by year-end 2015, with subject patients to be followed for three years. Interested persons may schedule an appointment to be screened for inclusion or request more information by contacting Crissie DeSpirito at 502-852-0026 or crissie.despirito(at)louisville.edu. Additional information on the LIBERATE study is available on the national clinical trials website, ClinicalTrials.gov, using the Clinical Trials Identifier NCT01796392 or by calling 1-888-248-LUNG.
For more information about EBV technology, visit: http://www.fda.gov/ohrms/dockets/ac/08/slides/2008-4405s1-03.pdf
Sources:
University of Louisville
U.S Food and Drug Administration
ClinicalTrials.gov
Image Crredits
University of Louisville
U.S Food and Drug Administration

I want to have this precudre done do you do the procudre or can I be in on clinical trial? Please let me know I am desperate.