 A team of researchers at INSERM (Institut national de la santé et de la recherche médicale), University of Bordeaux and Bordeaux University Hospital in France recently reported on the clinical efficacy of gallopamil, in the treatment of patients with severe asthma. The study was published in the American Journal of Respiratory and Critical Care Medicine and is entitled “Calcium Channel Blocker Reduces Airway Remodeling in Severe Asthma: a Proof-of-concept Study.”
A team of researchers at INSERM (Institut national de la santé et de la recherche médicale), University of Bordeaux and Bordeaux University Hospital in France recently reported on the clinical efficacy of gallopamil, in the treatment of patients with severe asthma. The study was published in the American Journal of Respiratory and Critical Care Medicine and is entitled “Calcium Channel Blocker Reduces Airway Remodeling in Severe Asthma: a Proof-of-concept Study.”
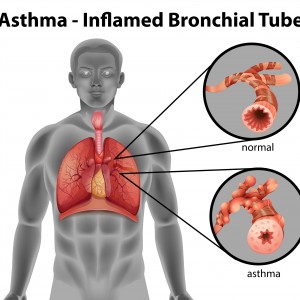
Asthma is a common chronic inflammatory lung disease characterized by airflow obstruction that causes wheezing (whistling sound when the person breathes), shortness of breath, chest tightness, and coughing. This respiratory disorder is thought to be caused by a combination of both genetic and environmental factors. In severe asthma, the obstruction of the airways is exacerbated and individuals experience persistent breathing difficulty, limited physical activity and asthma attacks. Severe asthma is a condition that affects 1 to 3% of the world’s population and is responsible for a significant number of emergency hospital admissions.
The bronchial obstruction observed in severe asthma results from a remodeling of the airways, namely by an increase of the bronchial smooth muscle (BSM) mass. This atypical BSM growth was found to be associated with resistance to intensive treatment and a poor prognosis, and is triggered by abnormal calcium entry into the BSM cells. Corticosteroids, which are one of the standard treatments for severe asthma, are unable to prevent the excessive proliferation of BSM cells.
Researchers had previously found that gallopamil, a calcium channel blocker usually prescribed for the treatment of cardiac arrhythmias, is capable of reducing BSM proliferation. In this study, a randomized, double-blind, placebo-controlled clinical trial was established to assess the in vivo efficacy of gallopamil in 31 patients with severe asthma. For a period of one year, data regarding asthma attacks, drug effects, BSM and bronchial wall thickness was collected. Patients were monitored for an additional three-month follow-up period without treatment.
Researchers found that gallopamil significantly reduced BSM in asthmatic patients in comparison with the placebo group, resulting in a considerable reduction in bronchial wall thickness. During the follow-up period, patients who had been given gallopamil reported fewer prolonged asthma attacks than individuals in the placebo group. No side effects were reported.
The team concluded that the calcium channel blocker is capable of reducing bronchial remodeling in severe asthma patients and prevent the occurrence of asthma exacerbations. Further studies to confirm the clinical efficacy of gallopamil should be conducted using larger patient cohorts and extended periods of treatment.

